Chapter 8 - Redox Reactions Exercise 272
Solution 1
a. NaH2PO4
Let the oxidation number of P be x.
We know that,
Oxidation number of Na = +1
Oxidation number of H = +1
Oxidation number of O = –2
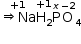
Then, we have
1(+1) +2(+1)+1(x)+4(-2) =0
 1+2+x-8 = 0
1+2+x-8 = 0
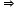 x = +5
x = +5
Hence, the oxidation number of P is +5.
b. NaHSO4
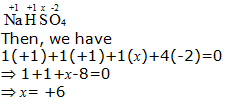
Hence, the oxidation number of S is + 6.
c. H4P2O7
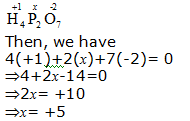
Hence, the oxidation number of P is + 5.
d. K2MnO4
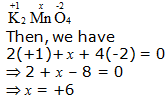
Hence, the oxidation number of Mn is + 6.
e. CaO2
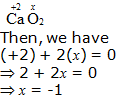
Hence, the oxidation number of O is – 1.
f. NaBH4
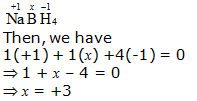
Hence, the oxidation number of B is + 3.
g. H2S2O7
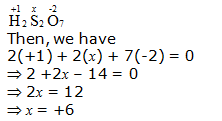
Hence, the oxidation number of S is + 6.
h. KAl(SO4)2.12H2O
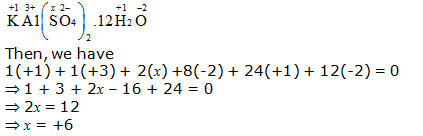
Or,
We can ignore the water molecule as it is a neutral molecule. Then, the sum of the oxidation numbers of all atoms of the water molecule may be taken as zero. Therefore, after ignoring the water molecule, we have
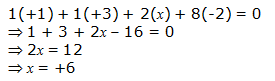
Hence, the oxidation number of S is + 6.
Let the oxidation number of P be x.
We know that,
Oxidation number of Na = +1
Oxidation number of H = +1
Oxidation number of O = –2
Then, we have
1(+1) +2(+1)+1(x)+4(-2) =0
Hence, the oxidation number of P is +5.
b. NaHSO4
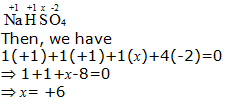
Hence, the oxidation number of S is + 6.
c. H4P2O7
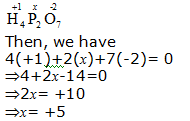
Hence, the oxidation number of P is + 5.
d. K2MnO4
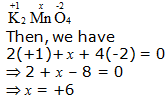
Hence, the oxidation number of Mn is + 6.
e. CaO2
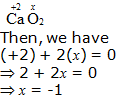
Hence, the oxidation number of O is – 1.
f. NaBH4
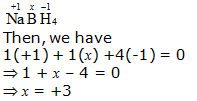
Hence, the oxidation number of B is + 3.
g. H2S2O7
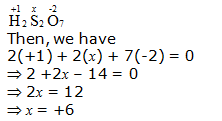
Hence, the oxidation number of S is + 6.
h. KAl(SO4)2.12H2O
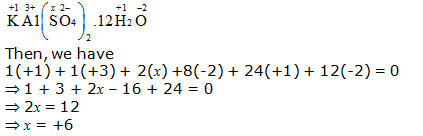
Or,
We can ignore the water molecule as it is a neutral molecule. Then, the sum of the oxidation numbers of all atoms of the water molecule may be taken as zero. Therefore, after ignoring the water molecule, we have
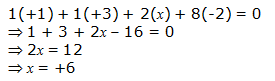
Hence, the oxidation number of S is + 6.
Solution 2
a. KI3
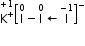
b. H2S4O6
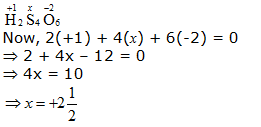
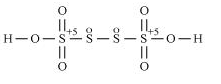
c. Fe3O4
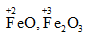 d. CH3CH2OH
d. CH3CH2OH
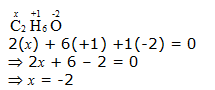
e. CH3COOH
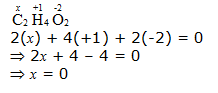
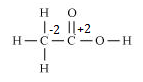
In KI3, the oxidation number (O.N.) of K is +1. Hence, the average oxidation number of I is 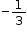 . However, O.N. cannot be fractional. Therefore, we will have to consider the structure of KI3 to find the oxidation states.
. However, O.N. cannot be fractional. Therefore, we will have to consider the structure of KI3 to find the oxidation states.
In a KI3 molecule, an atom of iodine forms a coordinate covalent bond with an iodine molecule.
Hence, in a KI3 molecule, the O.N. of the two I atoms forming the I2 molecule is 0, whereas the O.N. of the I atom forming the coordinate bond is –1.
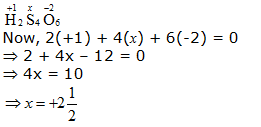
However, O.N. cannot be fractional. Hence, S must be present in different oxidation states in the molecule.
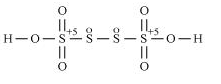
The O.N. of two of the four S atoms is +5 and the O.N. of the other two S atoms is 0.
On taking the O.N. of O as –2, the O.N. of Fe is found to be 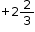 . However, O.N. cannot be fractional.
. However, O.N. cannot be fractional.
Here, one of the three Fe atoms exhibits the O.N. of +2 and the other two Fe atoms exhibit the O.N. of +3.
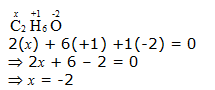
Hence, the O.N. of C is –2.
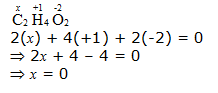
However, 0 is average O.N. of C. The two carbon atoms present in this molecule are present in different environments. Hence, they cannot have the same oxidation number. Thus, C exhibits the oxidation states of +2 and –2 in CH3COOH.
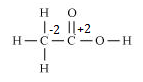
Solution 3

Solution 4

Solution 5

Solution 6
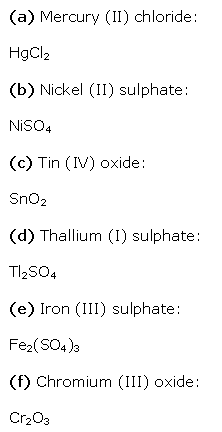
Solution 7


Solution 8

Solution 9

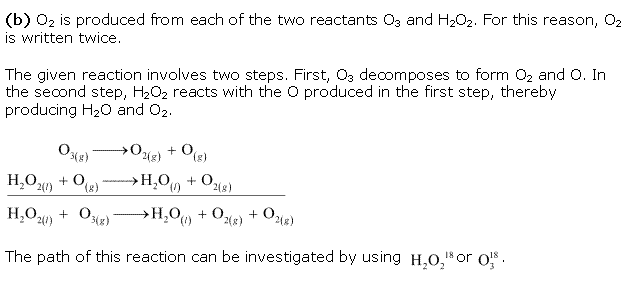
Chapter 8 - Redox Reactions Exercise 273
Solution 1


Solution 2

Solution 3

Solution 4

Solution 5
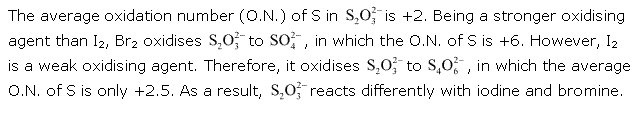
Solution 6

Solution 7
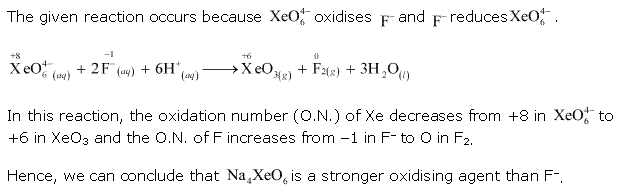
Solution 8

Chapter 8 - Redox Reactions Exercise 274
Solution 1



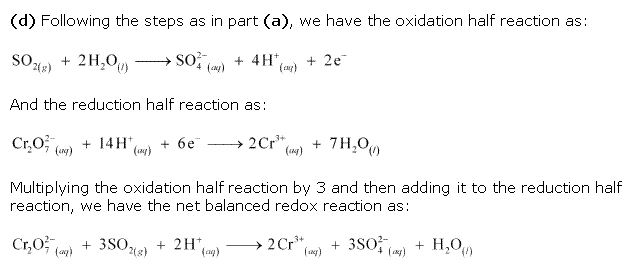
Solution 2






Solution 3

Solution 4

Solution 5

Solution 6

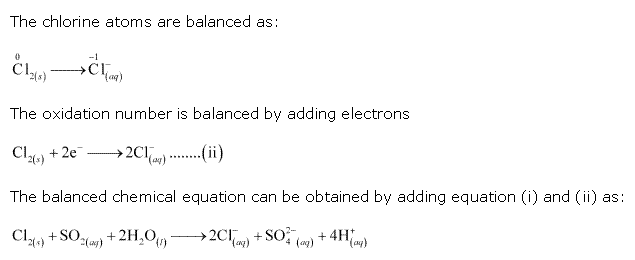
Solution 7

Solution 8

Solution 9

Chapter 8 - Redox Reactions Exercise 275
Solution 1

Solution 2

Solution 3
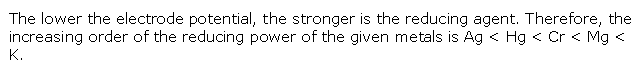
Solution 4

Comments
Post a Comment